ElectrochemistryHard
Question
The oxidation states of sulphide in S8, S2F2, H2S respectively, are
Options
A.0, +1 and -2
B.+2, +1 and -2
C.0, +1 and +2
D.-2, 1 and -2
Solution
Oxidation number of S in S8 = 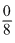 = 0
= 0
Let oxidation number of S in S2F2 be x
2x +2(- 1) = 0 x=+1
in H2S 2 + x = 0
x = - 2
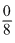 = 0
= 0Let oxidation number of S in S2F2 be x
2x +2(- 1) = 0 x=+1
in H2S 2 + x = 0
x = - 2
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A resistance of 50W is registered when two electrodes are suspended into a beaker containing a dilute solution of a stro...Two weak acid solutions HA1 and HA2 each with the same concentration and having pKa values 3 and 5 are placed in contact...The electrical conductivity of a solution serves as a means of determining the end point in a chemical reaction, involve...The limiting molar conductivities ∧o for NaCl, KBr and KCl are 126, 152 and 150 S cm2 mol-1 respectively. The W...Given EoCr3+/Cr = - 0.72V, EoFe2+/Fe = - 0.42 V. The potential for the cell Cr | Cr3+ (0.1M)||Fe2+(0.01M)|Fe is...