ElectrochemistryHard
Question
Two weak acid solutions HA1 and HA2 each with the same concentration and having pKa values 3 and 5 are placed in contact with hydrogen electrode (1 atm, 25oC) and are interconnected through a salt bridge. The emf of the cell is
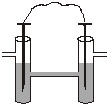
Options
A.0.21 V
B.0.059 V
C.0.018 V
D.0.021 V
More Electrochemistry Questions
Select the correct statement with respect tolocomotion in humans:Select the correct statement with respect tolocomotion ...The arrangement of X- ions around A+ ion in solid AX is given in the figure (not drawn to scale).If the radius of X- is ...A metallic rod of length ′l′ is tied to a string of length 2l and made to rotate with angular speed ω ...Zn Amalgam is prepared by electrolysis of aqueous ZnCl2 using Hg cathode (9gm). How much current is to be passed through...Consider the electrode Ag | AgCl(s), Cl-(0.1 M), i.e., silver electrode in contact with 0.1 M KCl solution saturated wit...