ElectrochemistryHard
Question
The electrical conductivity of a solution serves as a means of determining the end point in a chemical reaction, involved in the titration of acids, bases, or precipitation. Which of the following conductometric titrations represent the curve of HCl vs NH4OH
x - axis ⇒ Volume of alkali added
y - axis ⇒ Conductivity
x - axis ⇒ Volume of alkali added
y - axis ⇒ Conductivity
Options
A.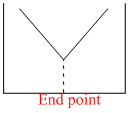

B.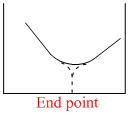

C.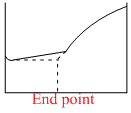

D.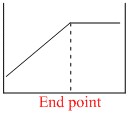

More Electrochemistry Questions
A 0.5 M NaOH solution offers a resistance of 31.6 ohm in a conducivity cell at roomtemperature. What shall be the appori...The emf of Daniel cell at 298 K is E1Zn|ZnSO4(0.01M)||CuSO4(1.0M)|Cu When the concentration of ZnSo4 is 1.0 M and that o...The molar conductivities ∧oNaOAc and ∧oHCl at infinite dilution in water at 25oC are 91.0 and 426.2 S cm2/mo...A hydrogen electrode is immersed in a solution with pH = 0 (HCl). By how much will the potential (reduction) change if a...In a galvanic cell...