ElectrochemistryHard
Question
Given EoCr3+/Cr = - 0.72V, EoFe2+/Fe = - 0.42 V. The potential for the cell
Cr | Cr3+ (0.1M)||Fe2+(0.01M)|Fe is
Cr | Cr3+ (0.1M)||Fe2+(0.01M)|Fe is
Options
A.0.26 V
B.0.399 V
C.- 0.339 V
D.- 0.26 V
Solution
As EoCr/Cr3+ = - 0.72 V and EoFe2+/Fe = - 0.42 V
2Cr + 3Fe2+ → 3Fe + 2Cr3+
Ecell = Eocell-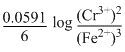
=(-0.42 + 0.72)-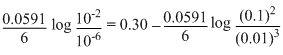
= 0.30 -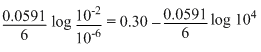
Ecell = 0.2606 V
2Cr + 3Fe2+ → 3Fe + 2Cr3+
Ecell = Eocell-
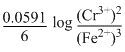
=(-0.42 + 0.72)-
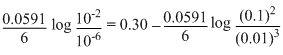
= 0.30 -
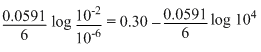
Ecell = 0.2606 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Calculate the solubility product of Co2[Fe(CN)6] in water at 25oC.Given, conductivity of saturated solutions of Co2[Fe(C...The electric charge for electrode deposition of one gram equivalent of a substance is :...The equivalent conductance of NaCl at concentration C and at infinite dilution are λC and λ∞ respectivel...Zinc granules are added in excess to a 500 ml of 1.0 M nickel nitrate solution at 25°C until the equilibrium is reached....Indicator electrode is...