ElectrochemistryHard
Question
A resistance of 50W is registered when two electrodes are suspended into a beaker containing a dilute solution of a strong electrolyte such that exactly half of the them are submerged into solution as shown in figure. If the solution is diluted by adding pure water (negligible conductivity) so as to just completely submerge the electrodes, the new resistance offered by the solution would be
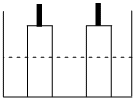

Options
A.50 Ω
B.100 Ω
C.25 Ω
D.200 Ω
Solution
R = 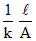 The k is halved while the A is doubled. Hence R remains 50 Ω.
The k is halved while the A is doubled. Hence R remains 50 Ω.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...Salt bridge contains :...The following electrochemical cell has been set up: Pt(s)|Fe3+, Fe2+ (a = 1)||Ce4+, Ce3+ (a = 1)|Pt(s); E°(Fe3+|Fe2+) = ...A current of 2 A was passed for 1 h through a solution of CuSO4 0.237g of Cu2+ ions were discharged at cathode. The curr...During the electrolysis of 0.1 M CuSO4 solution using copper electrodes, a depletion of [Cu++] occurs near the cathode w...