ElectrochemistryHard
Question
The equivalent conductances of two strong electrolytes at infinite dilution in H2O (where ions move freely through a solution) at 25oC are given below:
∧oCH3COONa = 91.0 S cm2 / equiv
∧oHCl 426.2 S cm2 / equiv
What additional information/quantity one needs to calculate ∧o of an aqueous solution of acetic acid?
∧oCH3COONa = 91.0 S cm2 / equiv
∧oHCl 426.2 S cm2 / equiv
What additional information/quantity one needs to calculate ∧o of an aqueous solution of acetic acid?
Options
A.∧o of NaCl
B.∧o of CH3COOK
C.The limiting equivalent conductance of H+ (∧oH+)
D.∧o of chloroacetic acid (C/CH2COOH)
Solution
From Kohlrausch′s law
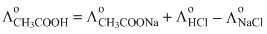
Hence, (1) is the correct answer.
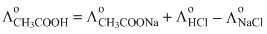
Hence, (1) is the correct answer.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Through a solution of CuSO4 a current of 3 amperes was passed for 2 hours. At cathode 3 g of Cu2+ ions were discharged. ...Zn + Cu2+(aq) ⇋ Cu + Zn2+(aq)Reaction quotient is Q = . Variation of Ecell with log Q is of the type with OA = 1.1...Chromium plating can involve the electrolysis of an electrolyte of an acidified mixture of chromic acid and chromium sul...If Fe3+ + Y4- ⇋ FeY-; Kf = 1.3 × 1025Fe2+ + Y4- ⇋ FeY2-; Kf = 2.1 × 1014and Fe3+ + e- ⇋ Fe2+...The potential of the Daniell cell, Zn Cu was reported by Buckbee, Surdzial and Metz as Eo = 1.1028 - 0.641 × 10-3 T...