ElectrochemistryHard
Question
Through a solution of CuSO4 a current of 3 amperes was passed for 2 hours. At cathode 3 g of Cu2+ ions were discharged. The current efficiency is [At. wt. of Cu = 63.5]
Options
A.33.3%
B.42.2%
C.48.7%
D.54.4%
Solution
According to law of electrolysis, Mass deposited (m) - Z i t
or i =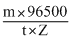
Here, m = 3g, t = 2 t = 2 × 60 × 60 = 7200sec
Z =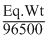 ; Eq. wt =
; Eq. wt = 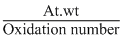
∴ i =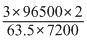 = 1.266A
= 1.266A
Efficiency of current
= × 100
× 100
=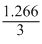 × 100 = 42.22%
× 100 = 42.22%
or i =
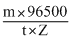
Here, m = 3g, t = 2 t = 2 × 60 × 60 = 7200sec
Z =
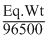 ; Eq. wt =
; Eq. wt = 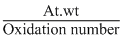
∴ i =
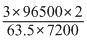 = 1.266A
= 1.266A Efficiency of current
=
 × 100
× 100=
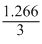 × 100 = 42.22%
× 100 = 42.22%Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The electrode potential becomes equal to standard electrode potential when reactants and products concentration ratio is...The electrode potential of electrode M(s) → Mn+ (aq) (2M) + ne-.at 298 K is E1. When temperature is doubled and co...The solubility product of silver iodide is 8.3 × 10-17 and the standard reduction potential of Ag, Ag+ electrode is...The standard reduction potential values of three metallic cations, X, Y and Z are +0.52, −3.03 and −1.18 V, respectively...A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...