ElectrochemistryHard
Question
Chromium plating can involve the electrolysis of an electrolyte of an acidified mixture of chromic acid and chromium sulphate. If during electrolysis the article being plated increases in mass by 2.6 g and 0.6 dm3 of oxygen are evolved at an inert anode, the oxidation state of chromium ions being discharged must be: (assuming atomic weight of Cr = 52 and 1 mole of gas at room temperature and pressure occupies a volume of 24 dm3)
Options
A.-1
B.Zero
C.+1
D.+2
Solution
4H+ → O2 + 2H2O + 4e- ⇒ 1 mol ≡ 4F
⇒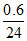 mol ≡ 4 ×
mol ≡ 4 × 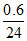 = 0.1F
= 0.1F
Cr3+ xe- → Cr(3 - x)+
⇒ 1 mol ≡ x F ⇒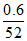 mol ≡
mol ≡ 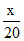 F ≡ 0.1 F ⇒ x = 2
F ≡ 0.1 F ⇒ x = 2
⇒ Oxidation state of Cr(3 - 2) = Cr+1 = + 1.
⇒
Cr3+ xe- → Cr(3 - x)+
⇒ 1 mol ≡ x F ⇒
⇒ Oxidation state of Cr(3 - 2) = Cr+1 = + 1.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
BY the electrolysis of aqueous solution of CuSO4, the products obtained at both the electrodes are...Which Shows electrical conductance ?...An ion is a reduced to the element when it absorbs 6 × 1020 electrons. The number of equivalents of the ion is...Which of the following statements is true for an electrochemical cell of Cu-H2....Equivalent conductance of an electrolyte containing NaF at infinite dilution is 90.1 Ohm-1 cm2 If NaF is replaced by KF ...