ElectrochemistryHard
Question
If Fe3+ + Y4- ⇋ FeY-; Kf = 1.3 × 1025
Fe2+ + Y4- ⇋ FeY2-; Kf = 2.1 × 1014
and Fe3+ + e- ⇋ Fe2+; E- = + 0.77 V
E- for Y- + e- → FeY2-
Fe2+ + Y4- ⇋ FeY2-; Kf = 2.1 × 1014
and Fe3+ + e- ⇋ Fe2+; E- = + 0.77 V
E- for Y- + e- → FeY2-
Options
A.0.13 V
B.- 0.636 V
C.+0.68 V
D.1.41 V
Solution
For FeY- → FeY2-, the change
Fe3+ + e- → Fe2+
E-Fe3+|FeY2|FeY-
E-Fe|Fe2+ +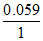 log
log 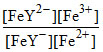
= 0.77 +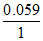 log
log 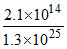 = 0.77 - 0.64 = 0.13V
= 0.77 - 0.64 = 0.13V
Fe3+ + e- → Fe2+
E-Fe3+|FeY2|FeY-
E-Fe|Fe2+ +
= 0.77 +
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...The current required to produce oxygen at the rate of 2.8 ml (0°C, 1 atm) per second during electrolysis of acidulated w...Copper sulphate solution (250 ml) was electrolysed using a platinum anode and a copper cathode. A constant current of 2 ...Using the standard potential values given below, decide which of the statements I, II, III, IV are correct. Choose the r...At 25°C, the solubility product of CuCl is 2.0 × 10−7 and $E_{Cl^{-}|CuCl|Cu}^{o}$ is 0.128 V. The value of $E_{Cu^{+}|C...