ElectrochemistryHard
Question
The potential of the Daniell cell, Zn 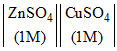 Cu was reported by Buckbee, Surdzial and Metz as Eo = 1.1028 - 0.641 × 10-3 T + 0.72 × 10-5 T2, where T is the temperature in degree celsius. Calculate ᐃSo for the cell reaction at 25oC :
Cu was reported by Buckbee, Surdzial and Metz as Eo = 1.1028 - 0.641 × 10-3 T + 0.72 × 10-5 T2, where T is the temperature in degree celsius. Calculate ᐃSo for the cell reaction at 25oC :
Options
A.- 45.32 EU
B.- 34.52 EU
C.- 25.43 EU
D.- 54.23 EU
Solution
Eo = 1.1028 - 0.641 × 10-3 T + 0.72 × 10-5 T2
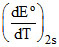 = - 0.641 × 10-3 + 2 × 0.72 × 10-5 T
= - 0.641 × 10-3 + 2 × 0.72 × 10-5 T
= (-0.641 + 0.36) × 10-3 = - 0.281 × 10-3
ᐃSo = nF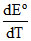 = 2 × 96500 × (-281 × 10-3) = - 54.23 EU
= 2 × 96500 × (-281 × 10-3) = - 54.23 EU
= (-0.641 + 0.36) × 10-3 = - 0.281 × 10-3
ᐃSo = nF
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
...The standard emf of the cell, Cd(s) | CdCl2(aq) (0.1 M) || AgCl(s) | Ag(s) in which the cell reaction is,Cd(s) + 2AgCl(s...For the process: Cu2+ + 2e− → Cu; log[Cu2+] vs. Ered graph is shown in the figure, where OA = 0.34 V. The electrode pote...In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...the variation of equivalent conductance of strong electrolyte with is correctly shown in the figure...