ElectrochemistryHard
Question
Zn + Cu2+(aq) ⇋ Cu + Zn2+(aq)
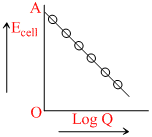
Reaction quotient is Q =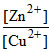 . Variation of Ecell with log Q is of the type with OA = 1.10 V. Ecell will be 1.1591 V when
. Variation of Ecell with log Q is of the type with OA = 1.10 V. Ecell will be 1.1591 V when
Reaction quotient is Q =

Options
A.[Cu2+] / [Zn2+] = 0.01
B.[Zn2+] / [Cu2+] = 0.01
C.[Zn2+] / [Cu2+] = 0.1
D.[Zn2+] / [Cu2+] = 1
Solution
Ecell = E-cell - 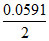 log
log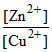
From line OA = E-cell = 1.10 V
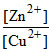 = 10-2 M, then Ecell = 1.1591 V
= 10-2 M, then Ecell = 1.1591 V
From line OA = E-cell = 1.10 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Through a solution of CuSO4 a current of 3 amperes was passed for 2 hours. At cathode 3 g of Cu2+ ions were discharged. ...For the process: Cu2+ + 2e− → Cu; log[Cu2+] vs. Ered graph is shown in the figure, where OA = 0.34 V. The electrode pote...The molar conductivity of NH4Cl, OH− and Cl− at infinite dilution is 150, 200 and 75 Ω−1 cm2 mol−1, respectively. If the...What is the pH of 0.01 m glycine solution ? for glycine, Ka1 = 4.5 × 10-3 and Ka2 = 1.7 × 10-10 at 298K ?...Although aluminium is above hydrogen in the electrochemical series, it is stable in air and water. Why?...