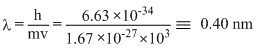Atomic StructureHard
Question
Calculate the wavelength (in nanometer) associated with a proton moving at 1.0 × 103ms-1
(Mass of proton = 1.67 × 10-27 kg and h = 6.63 × 10-34 js)
(Mass of proton = 1.67 × 10-27 kg and h = 6.63 × 10-34 js)
Options
A.0.032 nm
B.0.40 nm
C.2.5 nm
D.14.0 nm
More Atomic Structure Questions
The change in orbital angular momentum corresponding to an electron in Balmer transition inside a hydrogen atom can be...The energy required to break one mole of Cl-Cl bonds in Cl2 is 242 kJ mol-1. The longest wavelength of light capable of ...The angular momentum of electron in an excited H atom is . The P.E. of electron will be :...A bulb emits light of wavelength 1987 8 7.nm. The bulb is rated as 200 W and 14% of the energy is emitted as light. How ...When electron jumps from the fourth orbit to the second orbit in He+ ion, the radiation emitted out will fall in...