Atomic StructureHard
Question
The energy required to break one mole of Cl-Cl bonds in Cl2 is 242 kJ mol-1. The longest wavelength of light capable of breaking a single Cl - Cl bond is
(c = 3 × 108 ms-1 and NA = 6.02 × 1023 mol-1)
(c = 3 × 108 ms-1 and NA = 6.02 × 1023 mol-1)
Options
A.594 nm
B.640 nm
C.700 nm
D.494 nm
Solution
Energy required for 1 Cl2 molecule = 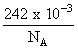 Joules
Joules
This energy is contained in photon of wavelength ′λ′
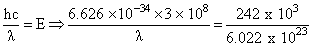
= 4947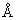 = 494 nm
= 494 nm
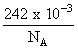 Joules
JoulesThis energy is contained in photon of wavelength ′λ′
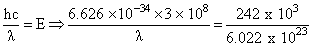
= 4947
 = 494 nm
= 494 nmCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
When KMnO4 acts as an oxidising agent and ultimately forms [MnO4]-1, MnO2, Mn2O3, Mn+2 then the number of electrons tran...The following charged particles accelerated from rest through the same potential difference are projected towards gold ...The number of radial nodes of 3s, 3p and 3d electrons are, respectively,...The ionization energy of H-atoms is 13.6 eV. The ionization energy of deuterium atom should be...The minimum uncertainty in de Broglie wavelength of an electron accelerated from rest by a potential difference of 6.0V...