Atomic StructureHard
Question
The change in orbital angular momentum corresponding to an electron in Balmer transition inside a hydrogen atom can be
Options
A.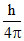
B.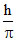
C.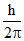
D.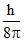
Solution
Change in angular momentum for 3 → 2 transition = (3 - 2) 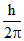 =
= 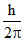
Change in angular momentum for 4 → 2 transition = (4 - 2)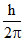 =
= 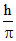 .
.
Change in angular momentum for 4 → 2 transition = (4 - 2)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The atomic nucleus contains :...What are the number of atoms per unit cell and the number of nearest neighbours in a body centered cubic structure?...If wavelength is equal to the distance travelled by the electron in one second, then -...The total number of orbitals in a shell with principal quantum number ′n′ is :...Find the frequency of light that corresponds to photons of energy 5.0 × 10-5 erg...