Atomic StructureHard
Question
The angular momentum of electron in an excited H atom is 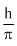 . The P.E. of electron will be :
. The P.E. of electron will be :
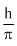 . The P.E. of electron will be :
. The P.E. of electron will be :Options
A.6.8 eV
B.3.4 eV
C.- 6.8eV
D.- 3.4 eV
More Atomic Structure Questions
Which of the following element will have the same number of electrons in s and as well as p-type of orbitals?...A photon of energy hn is absorbed by a free electron of a metal having work function w < hn. Then :...Wave number of the first line in the Balmer series of Be3+ is 2.5 × 105 cm−1. Wave number of the second line of the Pasc...The change in orbital angular momentum corresponding to an electron in Balmer transition inside a hydrogen atom can be...Sodium atoms emit a spectral line with a wavelength in the yellow, 589.6 nm. What is the approximate difference in ener...