Chemical Kinetics and Nuclear ChemistryHard
Question
For a first order reaction, (A) → products, theconcentration of A changes from 0.1 M to0.025 M in 40 minutes. The rate of reactionwhen the concentration of A is 0.01 M is :-
Options
A.1.73 × 10-4 M/min
B.1.73 × 10-5 M/min
C.3.74 × 10-4 M/min
D.3.74 × 10-5 M/min
Solution
For first order reaction
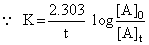
Given [A]0 = 0.1 M, [A]t = 0.025 M
∴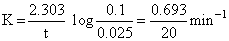
Rate = K [A]1
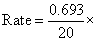 0.01 = 0.0003465 M/min
0.01 = 0.0003465 M/min
= 3.47 × 10-4 M/min
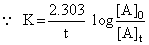
Given [A]0 = 0.1 M, [A]t = 0.025 M
∴
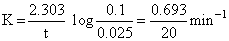
Rate = K [A]1
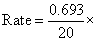 0.01 = 0.0003465 M/min
0.01 = 0.0003465 M/min= 3.47 × 10-4 M/min
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For a reaction A 2B starting with 1 M of ′A′ only, concentration of B (in M) after 100 sec. and 200 sec. is ...For the reaction2N2O5 → 4NO2 + O2The rate of reaction is :...Rate law of the reaction A → Product is, rate = k[A]. Graphically it is represented as Hence, rate constant is :...For the first order reaction :...Saponification of ethyl acetate (CH3COOC2H5) by NaOH (Saponification of ethyl acetate by NaOH is second order Rxn) is st...