Chemical Kinetics and Nuclear ChemistryHard
Question
Rate law of the reaction A → Product is, rate = k[A]. Graphically it is represented as
Hence, rate constant is :
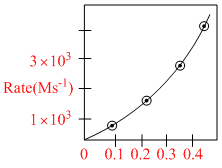
Hence, rate constant is :

Options
A. 3 × 10-4 s-1
B.1 × 10-2 s-1
C.3 × 10-2 s-1
D.1 × 10-4 s-1
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...The integrated rate equation is Rt = log C0 - log Ct. The straight line graph is obtained by plotting...In a certain reaction, 10% of the reactant decomposes in one hour, 20% in two hours, 30% in three hours, and so on. The ...In compounds of type ECl3 where E = B, P. As or Bi, the angles Cl − E − Cl for different R in order-...The standard enthalpies of formation of CO2(g), H2O(l) and glucose(s) at 250C are -400 kJ/mol, -300 kJ/mol & -1300 k...