Chemical Kinetics and Nuclear ChemistryHard
Question
Saponification of ethyl acetate (CH3COOC2H5) by NaOH (Saponification of ethyl acetate by NaOH is second order Rxn) is studied by titration of the reaction mixture initially having 1:1 molar ratio of the reactants. If 10 mL of 1 N HCl is required by 5 mL of the solution at the start and 8 mL of 1 N HCl is required by another 5 mL after 10 minutes, then rate constant is :
Options
A.k = 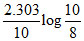
B.k = 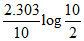
C.k = 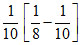
D.k = 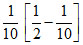
Solution
Saponification of ethyl acetate by NaOH is second order Rxn
Then k =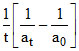
k =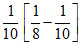
Then k =
k =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Milk is colloid in which :...Identify compounds readily liberate CO2 gas on heating ?...In a zero-order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10...The following mechanism has been proposed for the reaction of NO with Br2 to form NOBr: NO(g) + Br2(g) ⇋ NOBr2(g) ...the density of gold is 19g/cm3. If 1.9 × 10-4g of gold is dispersed in one litre of water to give a sol having sphe...