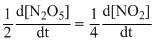Chemical Kinetics and Nuclear ChemistryHard
Question
For the reaction
2N2O5 → 4NO2 + O2
The rate of reaction is :
2N2O5 → 4NO2 + O2
The rate of reaction is :
Options
A.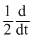 [N2O5]
[N2O5]
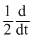 [N2O5]
[N2O5]B.2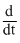 [N2O5]
[N2O5]
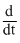 [N2O5]
[N2O5]C.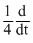 [NO2]
[NO2]
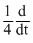 [NO2]
[NO2]D.4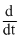 [NO2]
[NO2]
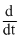 [NO2]
[NO2]Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If I is the intensity of absorbed light and C is the concentration of AB for photochemical process. AB + hv → AB*,...The correct increasing order of basic strength for the following compounds is :...Which of the following is not a green house gas?...Which of the following statements are correct about half-period....A catalyst lowers the activation energy of a reaction from 20 kJ mol-1 to 10 kJ mol-1. The temperature at which the unca...