Chemical Kinetics and Nuclear ChemistryHard
Question
For the first order reaction :
Options
A.the degree of dissociation is equal to (1 - e-kt)
B.a plot of reciprocal concentration of the reactant vs time gives a straight line
C.the time taken for the completions pf 75% reaction is thrice the 1/2 of the reaction
D.the pre-exponential factor in the Arrhenius equation has the dimension of time, T-1
Solution
For the first order reaction if degree of dissociation is α, then
K =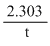 log
log 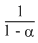
or k.t = 1oge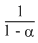 = - loge (1 - α)
= - loge (1 - α)
or e-kt = 1 - α
a = 1 - e-kt
According to Arrhenius equation
k = A.e-Ea/RT
Thup, dimension of A in the above expression is the same as that of rate constant, and for girst order reaction unit of k is time-1 (or T-1)
K =
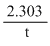 log
log 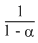
or k.t = 1oge
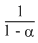 = - loge (1 - α)
= - loge (1 - α)or e-kt = 1 - α
a = 1 - e-kt
According to Arrhenius equation
k = A.e-Ea/RT
Thup, dimension of A in the above expression is the same as that of rate constant, and for girst order reaction unit of k is time-1 (or T-1)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A(g) → B(g) + (α - particles)(α-particles are helium nuclei, so will form helium gas by trapping electro...In general, the rate of a reaction can be increased by all the factors except...Match the graphical study with the order of the reactions : I : Rate II : Half life III : (a - x)-1...Which of the following is a lyophobiccolloidal solution?...The half-life of a first-order reaction is 10 minutes. In what time, the rate of reaction will decrease from 6.0 × 1021 ...