Chemical Kinetics and Nuclear ChemistryHard
Question
For a reaction A 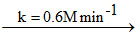 2B starting with 1 M of ′A′ only, concentration of B (in M) after 100 sec. and 200 sec. is respectively?
2B starting with 1 M of ′A′ only, concentration of B (in M) after 100 sec. and 200 sec. is respectively?
Options
A.2 and 4
B.1 and 2
C.2 and 3
D.None of these
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the parallel reactions $A\overset{\quad K_{1}\quad}{\rightarrow}B\text{ and }A\overset{\quad K_{2}\quad}{\rightarrow...The activation energy of a reaction is zero. The rate constant of the reaction...Which of the following quantity/quantities is/are temperature independent?...3A → B + CIt would be a zero order reaction when :...The order of reaction A → Products, may be given by which of the following expression(s)?...