Chemical Kinetics and Nuclear ChemistryHard
Question
The following statement (s) is (are) correct
Options
A.A plot of log Kpvs 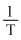 is linear
is linear
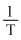 is linear
is linear B.A plot of log [X] vs time is linear for a first order reaction, x → P
C.A plot of P vs 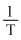 is linear at constant volume
is linear at constant volume
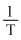 is linear at constant volume
is linear at constant volume D.A plot of P vs 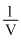 is linear at constant temperature
is linear at constant temperature
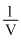 is linear at constant temperature
is linear at constant temperatureSolution
The relevant expressions are as follows :
(a) log Kp =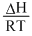 + Constant
+ Constant
(b) log [X] = log[X]0 + kt
(c)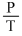 = Constant(V Constant)
= Constant(V Constant)
(d) PV = Constant (T Constant)
(a) log Kp =
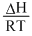 + Constant
+ Constant (b) log [X] = log[X]0 + kt
(c)
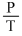 = Constant(V Constant)
= Constant(V Constant)(d) PV = Constant (T Constant)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In a zero order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10...For irreversible elementary reactions in parallel $A\overset{\quad K_{1}\quad}{\rightarrow}R\text{ and }A\overset{\quad ...During the kinetic study of the reaction, 2A + B → C + D, following results were obtained Based on the above data ...Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in fi...For a second order reaction plots are made for vs time for the reaction, 2A → Product. Pick up the correct sentenc...