Chemical Kinetics and Nuclear ChemistryHard
Question
During the kinetic study of the reaction, 2A + B → C + D, following results were obtained

Based on the above data which one of the following is correct ?

Based on the above data which one of the following is correct ?
Options
A.rate = k [A]2[B]
B.rate = k [A] [B]
C.rate = k [A]2[B]2
D.rate = k [A] [B]2
Solution
Let the order of reaction with respect to A is x and with respect to B is y. Thus,
rate = k [A]x [B]y
For the given cases,
(i) rate = k (0.1)x (0.1)y = 6.0 × 10-3
(ii) rate = k (0.3)x (0.2)y = 7.2 × 10-2
(iii) rate = k (0.3)x (0.40)y = 2.88 × 10-1
(iv) rate = k (0.4)x (0.1)y = 2.40 ×10-2
On dividing eq. (i) by (iv), we get
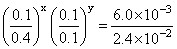
or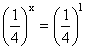
∴ x = 1
On dividing Eq. (ii) by (iii), we get
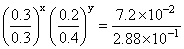
or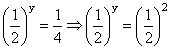
∴ y = 2
Thus, rate law is,
rate = k [A]1 [B]2 or
= k [A] [B]2
rate = k [A]x [B]y
For the given cases,
(i) rate = k (0.1)x (0.1)y = 6.0 × 10-3
(ii) rate = k (0.3)x (0.2)y = 7.2 × 10-2
(iii) rate = k (0.3)x (0.40)y = 2.88 × 10-1
(iv) rate = k (0.4)x (0.1)y = 2.40 ×10-2
On dividing eq. (i) by (iv), we get
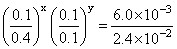
or
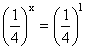
∴ x = 1
On dividing Eq. (ii) by (iii), we get
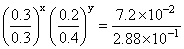
or
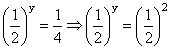
∴ y = 2
Thus, rate law is,
rate = k [A]1 [B]2 or
= k [A] [B]2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Given above is the concentration vs time plot for a dissociation reaction : $A \rightarrow nB$.Based on the data of the ...The rate of a reaction is approximately doubled for every 10°C rise in temperature. If the temperature is raised by 50°C...Which of the following pair of substances illustrates law of multiple proportions :-...What is the final product in the disintegrations series of starting from 92U235 :...The rate constant of a second-order reaction is 10−2 lit mole−1 s−1. The rate constant when expressed in ml molecule−1 m...