Chemical Kinetics and Nuclear ChemistryHard
Question
In a zero order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10oC to 100oC, the rate of the reaction will become
Options
A.256 times
B.512 times
C.64 times
D.128 times
Solution
For 10o rise in temperature, n = 1
so rate = 2n = 21 = 2
When temperature is increased from 10oC to 100oC, change in temperature = 100 - 10 = 90oC, i.e., n = 9. So rate = 29 = 512 times
Alternate methode With every 10o rise in temperature, rate becomes double, so
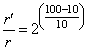 = 29 = 512 times.
= 29 = 512 times.
so rate = 2n = 21 = 2
When temperature is increased from 10oC to 100oC, change in temperature = 100 - 10 = 90oC, i.e., n = 9. So rate = 29 = 512 times
Alternate methode With every 10o rise in temperature, rate becomes double, so
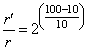 = 29 = 512 times.
= 29 = 512 times.Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
emits 8α - particles and 6β - particles. The netron /proton ratio in the product nucleus is :...X-Rays are emitted during...A(g) → B(g) + C(g) = k [A]At the start pressure is 100 mm and after 10 min , pressure is 120 mm. hence , rate cons...The rate constant k, for the reaction N2O5(g) → 2NO2(g) + O2(g) is 1.3 × 10-2s-1. Which equation given below ...Consider an endothermic reaction, X → Y with the activation energies Eb and Ef for the backward and forward reacti...