Chemical Kinetics and Nuclear ChemistryHard
Question
For a second order reaction plots are made for 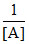 vs time for the reaction, 2A → Product. Pick up the correct sentences.
vs time for the reaction, 2A → Product. Pick up the correct sentences.
Options
A.the graph will show straight line with slope K
B.the graph will show straight line with intercept [A]0
C.the graph will show straight line with slope [A]0
D.the graph will show straight line with intercept 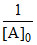
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Reaction A + B → C + D + 38 Kcal has activation energy 20 Kcal. Activation energy for the reaction C + D → A...Which of the following is incorrect about freundlich adsorption isotherm ?...The rate constant, the activation energy and the Arrhenious parameter of a chemical reaction at 25oC are 3.0 × 10-4...At a certain temperature, the reaction between NO and O2 to form NO2 is fast, while that between CO and O2 is slow. It m...In a consecutive reaction system $A\overset{\quad E_{1}\quad}{\rightarrow}B\overset{\quad E_{2}\quad}{\rightarrow}C$ whe...