Chemical Kinetics and Nuclear ChemistryHard
Question
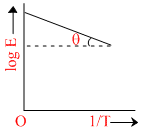
Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in figure if OX = 5 and slope of the line = - 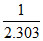 then Ea is :
then Ea is :

Options
A.2.303 × 2 cal
B.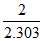 cal
cal
C.2 cal
D.None of these
Solution
We know, log K = log A - 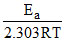
compare this by y = mx + c
m = -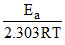 slope of this
slope of this
Given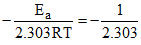
Ea = R = 2 cal
compare this by y = mx + c
m = -
Given
Ea = R = 2 cal
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A sedentary sea anemone gets attached to the shelllining of hermit crab. The association is :...Select correct statement (s) :...Order of reactivity is :-...A reaction is catalysed by H+ ion;and in the rate law the dependence of rate is of first order with respect to the conce...For a first order reaction, to obtain a positive slope we need to plot where [A] is theconcentration of reaction of reac...