Coordination CompoundHard
Question
Which of the following statements is correct?
Options
A.Geometrical isomerism is not observed in complexes of coordination number 4 having tetrahedral geometry
B.Square planar complexes generally do not show geometrical isomerism
C.The square planar complex of general formulae Ma3b or Mab3 exhibits cis-trans isomerism
D.The platinum glycinato complex, [Pt(gly)2] does not show geometrical isomerism
Solution
(A) The tetrahedral compounds can not show geometrical isomerism as we all know that all four positions are equivalent in tetrahedral geometry.
(B) In a square planar complex of formula [Ma2b2] [a and b are unidentate], the two ligands ′a′ may be arranged adjacent to each other in a cis isomer, or opposite to each other in a trans isomer.
(C) Ma3b and Mab3 have only one form.
(D)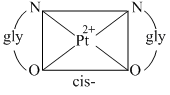
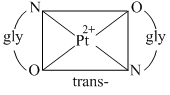
(B) In a square planar complex of formula [Ma2b2] [a and b are unidentate], the two ligands ′a′ may be arranged adjacent to each other in a cis isomer, or opposite to each other in a trans isomer.
(C) Ma3b and Mab3 have only one form.
(D)


Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is a high spin complex ?...The diamagnetic species is :...The value of the ′spin only′ magnetic moment for one of the following configurations is 2.84 BM. The correct...Which of the following complex will show optical activity ?...Which of the following does not have optical isomer ?...