Coordination CompoundHard
Question
Which of the following complex will show optical activity ?
Options
A.trans -[Co(NH3)4Cl2]+
B. [Cr(H2O)6]3+
C.cis -[Co(NH3)2(en)2]3+
D.trans -[Co(NH3)2(en)2]3+
Solution
(A) trans-[Co(NH3)4Cl2]+ has plane of symmetry as well as centre of symmetry. Hence it does not show optical isomerism.
(B) [Cr(H2O)6]3+ has plane of symmetry as well as centre of symmetry. Hence it does not show optical isomerism.
(C) (C) cis-[Co(NH3)2(en)2]3+ lacks one of the symmetry elements.
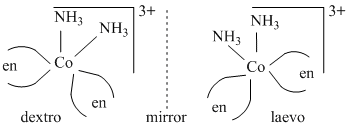
(D) trans-[Co(NH3)2(en)2]3+ has plane of symmetry as well as centre of symmetry. Hence it does not show optical isomerism.
(B) [Cr(H2O)6]3+ has plane of symmetry as well as centre of symmetry. Hence it does not show optical isomerism.
(C) (C) cis-[Co(NH3)2(en)2]3+ lacks one of the symmetry elements.

(D) trans-[Co(NH3)2(en)2]3+ has plane of symmetry as well as centre of symmetry. Hence it does not show optical isomerism.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which one is the most likely structure of CrCl3·6H2O if 1/3 of total chlorine of the compound is precipitated by adding ...Assign the hybridization, shape and magnetic moment of K3[Cu(CN)4] :...A complex compound of one cobalt (III) ion, two ′en′ molecules, two chloride ions and one nitrite ion will s...Which of the following pairs will show the same magnetic moment (′spin only′)?...The ionic radii of ð´+ and ðµ- ions are 0.98 ×10-10m and 1.81 ×10-10 m. The coordination number of each io...