Coordination CompoundHard
Question
Which of the following does not have optical isomer ?
Options
A.[Co(NH)3)3Cl3]
B.[Co(en)3)]Cl3
C.[Co(en)2)Cl2]Cl
D.[Co(en)(NH3)2)Cl2]Cl
Solution
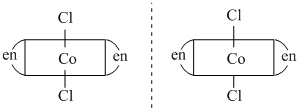
As clear from the figure, the mirror image is superimposable So, they are not differentcompounds.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which one of the following complexes in an outer orbital complex ?...Each of the following complex shows colour except...The ionic radii of ð´+ and ðµ- ions are 0.98 ×10-10m and 1.81 ×10-10 m. The coordination number of each io...Which statements is/are incorrect ?...In the coordination compound, K4[Ni(CN)4], the oxidation state of nickel is...