Coordination CompoundHard
Question
Which of the following is a high spin complex ?
Options
A.[Co(NH3)6]3+
B.[Fe(CN)6]4-
C.[Ni(CN)4]2-
D.[FeF6]3-
Solution
The complex [FeF6]4- is paramagnetic and uses outer orbital (4d) in hybridisation (sp3d2) ; it is thus called as outer orbital or high spin or spin free complex. So :
Fe2+, [Ar]3d6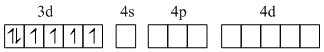
[FeF6]4-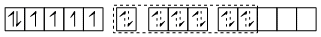
sp3d2 hybrid orbitals
Six pairs of electrons from six F- ions.
Fe2+, [Ar]3d6

[FeF6]4-
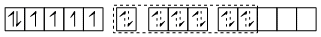
sp3d2 hybrid orbitals
Six pairs of electrons from six F- ions.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
A coordination compound of cobalt has the molecular formula containing five ammonia molecules, one nitro group and two c...The number of ions formed, when bis (ethane-1,2-diamine) copper (II) sulphate is dissolved in water will be...$\left\lbrack Ni\left( {PPh}_{3} \right)_{2}{Cl}_{2} \right\rbrack$ is a paramagnetic complex. Identify the INCORRECT st...The transition element which shows the highest oxidation state is :...Which of the following statements is/are incorrect for the complex [Cr(H2O)6]Cl3?...