Coordination CompoundHard
Question
The value of the ′spin only′ magnetic moment for one of the following configurations is 2.84 BM. The correct one is
Options
A.d4 (in strong ligand filed)
B.d4 (in weak ligand field)
C.d3 (in weak as well as in strong fields)
D.d5 (in strong ligand field)
Solution
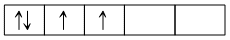
d4 in strong field, so unpaired electrons = 2.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The correct crystal field splitting and electron distribution is...Which is not a π-bonded complex ?...A complex of platinum, ammonia and chloride produces four ions per molecule in the solution. The structure consistent wi...The IUPAC name of Xe [Pt F6] is...What is the shape of Fe(CO)5 molecule ? Given that its dipole moment = 0....