Coordination CompoundHard
Question
The diamagnetic species is :
Options
A.[Ni(CN4)]2-
B.[NiCl4]2-
C.[NoCl4]2-
D.[NoF6]2-
Solution
In [Ni(CN4)]2-, nickel has oxidation no. of +2. So electronic configuration of its outer most orbital is as follows :
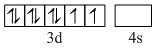
However,CN- is a strong ligand which forces the electron to pair up. Thus two unpaired electron are forced to pair up by CN-
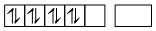
So, it becomes diamagnetic.
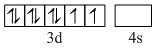
However,CN- is a strong ligand which forces the electron to pair up. Thus two unpaired electron are forced to pair up by CN-
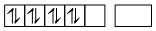
So, it becomes diamagnetic.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The compound Na2IrCl6 reacts with triphenylphosphine in diethyleneglycol in an atmosphere of CO to give [IrCl(CO)(PPh3)2...Wilkinson′s catalyst react with H2 to form an octahedral complex in which Rh (Z = 45) has the following electronic...The total number of possible coordination isomer for the given compound is[Pt(NH3)4Cl2][PtCl4]....Some salts although containing two different metallic elements give test for one of them in solution. Such salts are...Which one of the following statements is incorrect?...