Chemical Kinetics and Nuclear ChemistryHard
Question
For an endothermic reaction, energy of activation is Ea and enthalpy of reaction is ᐃH (both of these in KJ/mol). Minimum value of Ea will be
Options
A.less then ᐃH
B.equal to ᐃH
C.more then ᐃH
D.equal to zero
Solution
In endothermic reactions, energy of reactants is less than that of the products.
Potential energy diagram for endothermic reactions is,
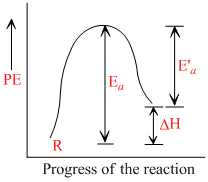
where, Ea = activation energy of backward reaction
E′a = activation energy of backward reaction
ᐃH = enthalpy of the reaction
From the above diagram,
Ea = E′a + ᐃH
Thus, Ea >ᐃH
Potential energy diagram for endothermic reactions is,

where, Ea = activation energy of backward reaction
E′a = activation energy of backward reaction
ᐃH = enthalpy of the reaction
From the above diagram,
Ea = E′a + ᐃH
Thus, Ea >ᐃH
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction wi...The compound A decomposed (A → product) by first order kinetics. If the concentration of A dropped from 0.1 M to 0...A substance ″A″ decomposes in solution following the first order kinetics. Flask I contains 1 L of 1 M solut...Activation energy (Ea) and rate constants (k1 and k2) of a chemical reaction of two differenttemperatures (T1 and T2) ar...