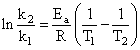Chemical Kinetics and Nuclear ChemistryHard
Question
Activation energy (Ea) and rate constants (k1 and k2) of a chemical reaction of two differenttemperatures (T1 and T2) are related by:
Options
A.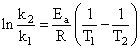
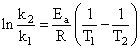
B.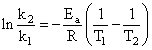
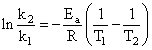
C.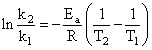
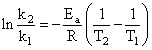
D.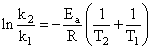
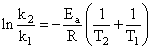
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Select the correct statement(s) -...For a given reaction, the concentration of the reactant plotted against time gave a straight line with negative slope. T...The composition of N2O5 is a first order reaction represented by: N2O5 → N2O4 + 1/2O2.After 20 min the volume of O...Inversion of sucrose (C12H22O11) is first-order reaction and is studied by measuring angle of rotation at different inst...For the first-order reaction A(g) → 2B(g) + C(g), the total pressure after time t from the start of reaction with A is P...