Chemical Kinetics and Nuclear ChemistryHard
Question
For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If 1.0 mole each of A and B introduced into a 1.0 L vessel and the initial rate was 1.0 × 10-2 mol L-1 s-1, rate when half reactants have been turned into products is :
Options
A.1.25 × 10-3 mol L-1 s-1
B.1.0 × 10-2 mol L-1 s-1
C.2.50 × 10-3 mol L-1 s-1
D.2.0 × 10-2 mol L-1 s-1
Solution
A + B → Product
Initial conc. 1 1
Initially
1.0 × 10-2 = K [1] [1]2
K = 1.0 × 10-2|
When half of reactant reacted
Rate = 1.0 × 10-2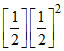
Rate = 1.25 × 10-3
Initial conc. 1 1
Initially
1.0 × 10-2 = K [1] [1]2
K = 1.0 × 10-2|
When half of reactant reacted
Rate = 1.0 × 10-2
Rate = 1.25 × 10-3
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Under the same reaction conditions, initial concentration of 1.386 mol dm-3 of a substance becomes half in 40 seconds an...The correct order of increasing basic nature for the bases NH3, CH3NH2 and (CH3)2NH is...The inversion of cane sugar proceeds with half life of 500 min at pH 5 for any concentration of sugar. However, if pH = ...For the reaction N2(g) + 3H2(g) → 2NH3(g) under certain conditions of temperature and partial pressure of the reactants,...Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrheni...