Chemical Kinetics and Nuclear ChemistryHard
Question
A substance ″A″ decomposes in solution following the first order kinetics. Flask I contains 1 L of 1 M solution of A and flask II contains 100 mL of 0.6 M solution. After 8 hr. the concentration, of A in flask I becomes 0.25 m. What will be the time for concentration of A in flask II to becomes 0.3 M ?
Options
A.0.4 hr
B.0.4 hr 2.4 hr
C.4.0 hr
D.Unpredictable as rate constant is not given
Solution
Flask I:
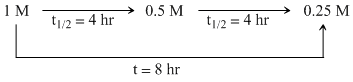
t1/2 = 4 hr.
Flask II:
0.6 M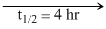 0.3 M
0.3 M
A first order reaction is independent of the initial concentration of the reactant.
Hence, answer is (c).
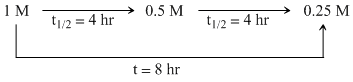
t1/2 = 4 hr.
Flask II:
0.6 M
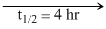 0.3 M
0.3 MA first order reaction is independent of the initial concentration of the reactant.
Hence, answer is (c).
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In compounds of type ECl3 where E = B, P. As or Bi, the angles Cl − E − Cl for different R in order-...For the parallel reactions $A\overset{\quad K_{1}\quad}{\rightarrow}B\text{ and }A\overset{\quad K_{2}\quad}{\rightarrow...Incorrect order of solubility is :...Units of rate constant of first and zero order reactions in terms of molarity M unit are respectively...In the reaction : P + Q →R + S the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. ...