Chemical Kinetics and Nuclear ChemistryHard
Question
The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction will be
(R = 8.314 JK-1 mol-1 and log 2 = 0.301)
(R = 8.314 JK-1 mol-1 and log 2 = 0.301)
Options
A.53.6 kJ mol-1
B.48.6 kJ mol-1
C.58.5 kJ mol-1
D.60.5 kJ mol-1
Solution


∴


∴
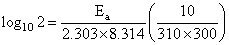
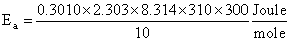
= 53.598 kJ/mol
Ea = 53.6 kJ/mol.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...The rate constant for the reaction, 2N2O5 → 4NO2 + O2 is 3.0 × 10-4s-1. If start made with 1.0 mol L-1 of N2O...The Golgi complex plays a major role :...The correct increasing order of basic strength for the following compounds is :...β - particle is emitted in radioactivity by...