Chemical Kinetics and Nuclear ChemistryHard
Question
The compound A decomposed (A → product) by first order kinetics. If the concentration of A dropped from 0.1 M to 0.05 M in 30 minutes, find the rate of reaction after 60 minutes, from start in M min-1
Options
A.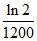
B.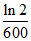
C.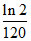
D.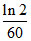
Solution
From given data t1/2 = 30 minute
K =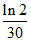
rt = 60 = k[A]t = 60
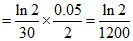
K =
rt = 60 = k[A]t = 60
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If no catalyst (H+) is present in acid hydrolysis of ester (in above question) then rate constant K is :...The correct order of increasing basic nature for the bases NH3, CH3NH2 and (CH3)2NH is...The rate of a chemical reaction doubles for every 10oC rise of temperature. If the temperature is raised by 50oC, the ra...Identify compounds readily liberate CO2 gas on heating ?...Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrheni...