Chemical Kinetics and Nuclear ChemistryHard
Question
The standard enthalpies of formation of CO2(g), H2O(l) and glucose(s) at 250C are -400 kJ/mol, -300 kJ/mol & -1300 kJ/mol respectively. The standard enthalpy of combustion per gram of glucose at 250C is -
Options
A.+ 2900 kJ
B.- 2900 kJ
C.- 16.11 kJ
D.+ 16.11 kJ
Solution
Combustion reaction for glucose is
C6H12O6(s) + 6O2(g)→ 6CO2(g) + 6H2O(l)
áƒHr = 6(-400) + 6(-300) - (-1300)
= -2900 KJ/mol 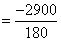 KJ / gm
KJ / gm
= -16.11 KJ/gm
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The following mechanism has been proposed for the reaction of NO with Br2 to form NOBr: NO(g) + Br2(g) ⇋ NOBr2(g) ...The differential rate law for the reaction H2 + I2 → 2HI is...The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration ...Which of the following graphs represents zero order if A → PAt t = 0 ⇒ [A]0At t = 0 ⇒ [A]t...A reaction of first-order completed 90% in 90 minutes, hence, it is completed 50% in approximately :...