Chemical Kinetics and Nuclear ChemistryHard
Question
The following mechanism has been proposed for the reaction of NO with Br2 to form NOBr:
NO(g) + Br2(g) ⇋ NOBr2(g)
NOBr2(g) + NO(g) → 2NOBr(g)
If the second step is the rate determining step, the order of the reaction with respect to NO(g) is
NO(g) + Br2(g) ⇋ NOBr2(g)
NOBr2(g) + NO(g) → 2NOBr(g)
If the second step is the rate determining step, the order of the reaction with respect to NO(g) is
Options
A.1
B.0
C.3
D.2
Solution
NO(g) + Br2(g) ⇋ NOBr2(g)
NOBr2(g) + NO(g) → 2NOBr(g)
R = K[NOBr2] [NO]
= K. K [NO][Br2][NO], where Kc =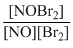
= K′[NO]2 [Br2]
NOBr2(g) + NO(g) → 2NOBr(g)
R = K[NOBr2] [NO]
= K. K [NO][Br2][NO], where Kc =
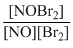
= K′[NO]2 [Br2]
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The first order rate constant for a certain reaction increase from 1.667 × 10-6 s-1 at 727oC to 1.66 × 10-4s-1...Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure. The corr...Which of the following statements are true for chemisorption...The rate of a first-order reaction is 0.04 mol l-1 s-1 at 10 second and 0.03 mol l-1 s-1 at 20 seconds afterinitiation o...Inversion of sucrose (C12H22O11) is first-order reaction and is studied by measuring angle of rotation at different inst...