Chemical Kinetics and Nuclear ChemistryHard
Question
The differential rate law for the reaction H2 + I2 → 2HI is
Options
A.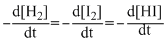
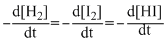
B.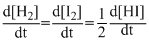
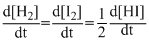
C.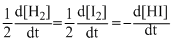
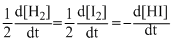
D.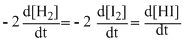
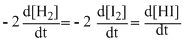
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The cell-mediated immunity inside the human bodyis carried out by :...Following are the values of Ea and DH for three reactions carried out at the same temperature : I : Ea = 20 kJ mol-1, &#...The reaction 2N2O5(g) → 4NO2(g) + O2(g) follows first order kinetics. The pressure of a vessel containing only N2O...The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...for a reaction A → B, Ea = 10 kJ mol-1, ᐃH = 5 kJ mol-1. Thus, potential energy profile for this reaction is...