Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following graphs represents zero order if
A → P
At t = 0 ⇒ [A]0
At t = 0 ⇒ [A]t
A → P
At t = 0 ⇒ [A]0
At t = 0 ⇒ [A]t
Options
A.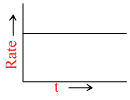

B.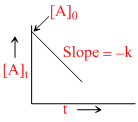

C.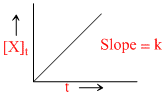

D.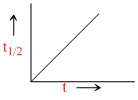

Solution
t1/2 ∝ (a)1-n; [A]t = [A]0 - kt and X = kt are integrated from of zero order.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following is not a green house gas?...For the reaction2N2O5 → 4NO2 + O2The rate of reaction is :...Read the following industrial methods for the preparation of H2SO4 and answer the question at the end. Professor Molina ...For an endothermic reaction, energy of activation is Ea and enthalpy of reaction is ᐃH (both of these in KJ/mol). ...Activation energy (Ea) and rate constants (k1 and k2) of a chemical reaction of two differenttemperatures (T1 and T2) ar...