Chemical Kinetics and Nuclear ChemistryHard
Question
The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration of A and halving the concentration of B, the ratio of the new rate to the earlier rate of the reaction will be as :
Options
A.1/2m + n
B.(m + n)
C.(n - m)
D.2(n - m)
Solution
Rate = K[A]n [B]m
Given, doubling the concentration of A and halving the concentration of B
then Rate2 = K[2A]n [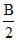 ]m
]m
Rate2 = K[A]n [B]mx 2(n-m)
Given, doubling the concentration of A and halving the concentration of B
then Rate2 = K[2A]n [
Rate2 = K[A]n [B]mx 2(n-m)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A stable hydrate :-′A′ may be :-...In the transformation of if one emission is an α-particle, what should be the other emission(s)?...The rate of a reaction A + 2B → P is 2 × 10−2 M/ min, when concentrations of each A and B are 1.0 M. If the rate of reac...β - particle is emitted in radioactivity by...When initial concentration of the reactant is doubled, the half-life period of a zero order reaction...