Chemical Kinetics and Nuclear ChemistryHard
Question
A reaction of first-order completed 90% in 90 minutes, hence, it is completed 50% in approximately :
Options
A.50 min
B.54 min
C.27 min
D.62 min
Solution
K = 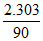 log
log 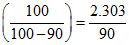 × log10 =
× log10 = 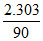 min-1
min-1
t1/2 (50% completed) =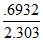 × 90 = 27.08 minutes.
× 90 = 27.08 minutes.
t1/2 (50% completed) =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If the rate of a gaseous reaction is independent of partial pressure of reactant, then the order of reaction is...At a certain temperature, the reaction between NO and O2 to form NO2 is fast, while that between CO and O2 is slow. It m...T50 (Half-life period) of first-order reaction is 10 minute. Starting with 10 mol L-1, rate after 20 minute is :...For the reaction A + 2B → C, rate is given by R = [A] [B]2 then the order of the reaction is...A stable hydrate :-′A′ may be :-...