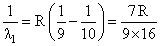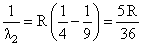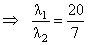Atomic StructureHard
Question
Electron in hydrogen atom first jumps from third excited state to second excited state and then fromsecond excited to the first excited state. The ratio of the wavelengths λ1 : λ2 emitted in the two casesis
Options
A.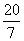
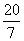
B.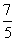
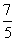
C.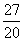
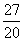
D.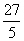
More Atomic Structure Questions
Two hydrogen atoms are in excited state with electrons residing in n = 2. First one is moving towards left and emits a p...The electron in a hydrogen atom makes a transition from an excited state to the ground state. Which of the following sta...A neutron makes a head-on elastic collision with a stationary deuteron. The fractional energy loss of the neutron in the...The potential difference applied to an X-ray tube is 5 kV and the current through it is 3.2 mA. The number of electrons ...The de-Broglie wavelength associated with the electron in the n = 4 level is :...