Atomic StructureHard
Question
The de-Broglie wavelength associated with the electron in the n = 4 level is :
Options
A.1/4th of the de-Broglie wavelength of the electron in the ground state.
B.four times the de-Broglie wavelength of the electron in the ground state
C.two times the de-Broglie wavelength of the electron in the ground state
D.half of the de-Broglie wavelength of the electron in the ground state
Solution
De-Broglie wavelength of electron
λ =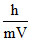 V ∝ 1/n
V ∝ 1/n
λ ∝ n
λ4 = 4λ1
∴ option (2)
λ =
λ ∝ n
λ4 = 4λ1
∴ option (2)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The potential difference applied to an X-ray tube is 5 kV and the current through it is 3.2 mA. The number of electrons ...The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in...If elements which principle quantum number n > 4 were not allowed in nature, the number of possible elements would be...Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of necleus and M2 the mass of nucleus. Then...If the electron in hydrogen orbit jumps from third orbit to second, the wavelength of the emitted radiation is given by ...