SolutionHard
Question
At particular temperature, the vapour pressure of two liquids A and B are respectively 120 and 180 mm of mercury. If 2 moles of A and 3 moles of B are mixed to form an ideal solution, at the vapour pressure of the solution at the same temperature will be (in mm of mercury)
Options
A.156
B.145
C.150
D.108
Solution
Vapour pressure of solution = PA + PB
= pAoxA + PBoxB =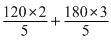 = 156 mm Hg
= 156 mm Hg
= pAoxA + PBoxB =
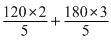 = 156 mm Hg
= 156 mm HgCreate a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of a solvent is decreased by 10 mm of Hg when a non-volatile solute was added to the solvent. The mo...A beaker (A) contains 20 g sugar in 100 g water. Another beaker (B) contains 10 g of sugar in 100 g water. Both the beak...The vapour pressure of a solution of a non-volatile solute B in a solvent A is 95% of the vapour pressure of the solvent...An ideal mixture of liquids A and B with 2 moles of A and 2 moles of B has a total vapour pressure of 1 atm at a certain...If 10-4 dm3 of water is introduced into a 1.0 dm3 flask to 300 K, how many moles of water are in the vapour phase when e...