SolutionHard
Question
The vapour pressure of a solution of a non-volatile solute B in a solvent A is 95% of the vapour pressure of the solvent at the same temperature. If the molecular weight of the solvent is 0.3 times the molecular weight of the solute, what is the ratio of weight of solvent to solute.
Options
A.0.15
B.5.7
C.0.2
D.none of these
Solution
According to question Ps = 0.95 P0
according raoul′s law Ps = P0 XA given MA = 0.3 MB
0.95 P0 = P0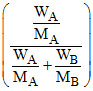 MA = molecular wt. of solvent
MA = molecular wt. of solvent
MB = molecular wt. of solute
0.95 =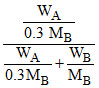 WA = gram wt. of solvent
WA = gram wt. of solvent
WB = gram wt. of solute
on solving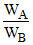 = 5.7.
= 5.7.
according raoul′s law Ps = P0 XA given MA = 0.3 MB
0.95 P0 = P0
MB = molecular wt. of solute
0.95 =
WB = gram wt. of solute
on solving
Create a free account to view solution
View Solution FreeMore Solution Questions
A teacher one day pointed out to his students the preculia fact that water is unique liquid which freezes exactly at 0o ...At a particular temperature and at a pressure of 1 atm, the mass and volume of O3 that can be dissolved in 1 L of H2O is...The cell constant of a given cell is 0.47 cm-1. The resistance of a solution placed in the cell is measured to be 31.6 o...Identify the false statement :...If vapour pressures of pure liquids ′A′ & ′B′ are 300 and 800 torr respectively at 25oC. When th...