SolutionHard
Question
If 10-4 dm3 of water is introduced into a 1.0 dm3 flask to 300 K, how many moles of water are in the vapour phase when equilibrium is established ?
(Given : Vapour pressure of H2O at 300 K is 3170 Pa ; R = 8.314 J K-1 mol-1)
(Given : Vapour pressure of H2O at 300 K is 3170 Pa ; R = 8.314 J K-1 mol-1)
Options
A.5.56 × 10-3 mol
B.1.53 × 10-2 mol
C.4.46 × 10-2 mol
D.2.27 × 10-3 mol
More Solution Questions
The freezing point depression of 0.001 m Kx[Fe(CN)6] is 7.10 × 10-3 K. Determine the value of x ? (Given Kƒ = ...Molal depression constant for Pb is 8.5°C kg/mol. How many grams of Sn must be dissolved in 425 g of Pb to produce an al...Which statement comparing solutions with pure solvent is not correct...Select correct statement :...When 25 ml of CCl4 and 25 ml of toluene is mixed, the total volume of the solution will be...
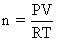 = 128 × 10-5 moles
= 128 × 10-5 moles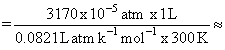 127 × 10-3 mol
127 × 10-3 mol