Ionic EquilibriumHard
Question
A + 2B ⇋ 2C + D, concentration of B was 1.5 time that of A, but the equilibriumconcentration of A and B are found to be equal. The equilibrium constant for the reaction is
Options
A.4
B.8
C.12
D.16
Solution
A + 2B ⇋ 2C + D
Intial 1 1.5 0 0
Equb 1-x 1.5 - 2x 2x x
Atequilibrium, [A] = [B]
1 - x = 1.5 - 2x
x = 0.5
∴ KC =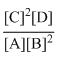
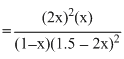
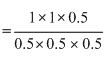 = 4
= 4
Intial 1 1.5 0 0
Equb 1-x 1.5 - 2x 2x x
Atequilibrium, [A] = [B]
1 - x = 1.5 - 2x
x = 0.5
∴ KC =
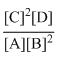
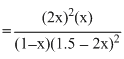
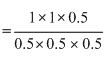 = 4
= 4Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility product of PbI2 is 7.2 × 10−9. The maximum mass of NaI which may be added in 500 ml of 0.005 M-Pb(NO3)2 s...Which is/are correct statements : (a) When 100 ml of 0.1 M NaCN solution is titrated with 0.1 M HCl solution the variati...Which of the following when mixed, will give a solution with pH > 7....Which statement is/are correct:...20 cm3 of x M solution of HCl is exactly neutralised by 40 cm3 of 0.05 M NaOH solution, the pH of HCl solution is...